📖 Learning

History taking 📝
Clinical examination 🔍
Vivas and Spotters 📚
Acutely Unwell Patients 🚨
Clinical Procedures 🏥
Interpretation 🔍
Prescribing 💊
Communication 🗣️
Clerking and reviewing 📋
🤔 Users completed 8 notes yesterday
🏥 Stations

🏥 Stations available: 207
Medicine 💊
Cardiology 🫀
Respiratory 🫁
Gastrointestinal 🍽️
Neurology 🧠
General surgery 🔪
ENT 👂
Orthopaedics 🦴
Urology 💧
Vascular 🩸
Dermatology 🌞
O&G 👶
Child health 🧸
Psychiatry 🧩
Sexual health 🔥
💁♂️ users completed 5 stations yesterday
🧠 Qbank

4 main parts of the site

1. MLA questions 🫀: Mapped to the MLA curriculum

2. Taylor francis 🧠: over 2500+ questions licenced from 18 text-books worth £191

3. Past examiners 🫁: Questions written by previous Medical School examiners

4. Track your performance 🏃♀️: QBank uses intelegent software to keep you on track
The reviews are in
Don't take our word for it


"The stations you provide are strikingly similar to those I came across during my medical school finals (some even verbatim!), and I have tried many other exam platforms. I'm truly grateful for your priceless support throughout my final couple of years at medical school!"
Raza Q 🇬🇧
"It has absolutely everything for medical school, so many histories with detailed differential diagnoses, how to approach emergencies, commonly prescribed drugs..every kind go examination you’ll ever need in osces"
John R 🇬🇧
"Thank you SO MUCH for the amazing educational resource. I’ve tried lots of platforms and books with mock OSCE stations and yours is by far and away the best I’ve tried"
Ed M 🇳🇿
"Get this right away. So helpful for OSCEs but also general clinical learning and understanding. Wish I had brought it sooner"
Emma W 🇬🇧
"Without a doubt, your platform outshines all other OSCE resources currently available. In all honesty, I can confidently attribute my success in securing a distinction in my finals to OSCEstop."
Harish K 🇬🇧
"OSCEstop distinguishes itself from many other platform banks by offering a wealth of questions that mimic the demanding and complex aspects of our finals. This platform played a crucial role in ensuring I was ready for the level of difficulty that awaited me in my final exams."
Natasha W 🇳🇿
🫀 Conditions

Cardiovascular 🫀
Gastrointestinal 🍽️
Neurology 🧠
Respiratory 🫁
Renal 🫙
General surgery 🔪
Urology 💧
ENT 👂
Vascular 🩸
Child health 🧸
Dermatology 🌞
Psychiatry 🧩
O&G 👶
Anaesthetics 💤
🤔 Users completed 8 notes yesterday
Trending Conditions
📖 Book
😃 More

💳 Pricing

Membership includes access to all 4 parts of the site:

1. Learning 📖: All notes, viva questions, track progress

2. Stations 🏥: 10 years of past medical school stations. Includes: heart murmurs, ECGs, ABGs, CXR

3. Qbank 🧠: 2500+ questions from Taylor Francis books, complete MLA coverage

4. Conditions 🫀: all conditions mapped to MLA, progress tracking
The reviews are in
Don't take our word for it


"The stations you provide are strikingly similar to those I came across during my medical school finals (some even verbatim!), and I have tried many other exam platforms. I'm truly grateful for your priceless support throughout my final couple of years at medical school!"
Raza Q 🇬🇧
"It has absolutely everything for medical school, so many histories with detailed differential diagnoses, how to approach emergencies, commonly prescribed drugs..every kind go examination you’ll ever need in osces"
John R 🇬🇧
"Thank you SO MUCH for the amazing educational resource. I’ve tried lots of platforms and books with mock OSCE stations and yours is by far and away the best I’ve tried"
Ed M 🇳🇿
"Get this right away. So helpful for OSCEs but also general clinical learning and understanding. Wish I had brought it sooner"
Emma W 🇬🇧
"Without a doubt, your platform outshines all other OSCE resources currently available. In all honesty, I can confidently attribute my success in securing a distinction in my finals to OSCEstop."
Harish K 🇬🇧
"OSCEstop distinguishes itself from many other platform banks by offering a wealth of questions that mimic the demanding and complex aspects of our finals. This platform played a crucial role in ensuring I was ready for the level of difficulty that awaited me in my final exams."
Natasha W 🇳🇿
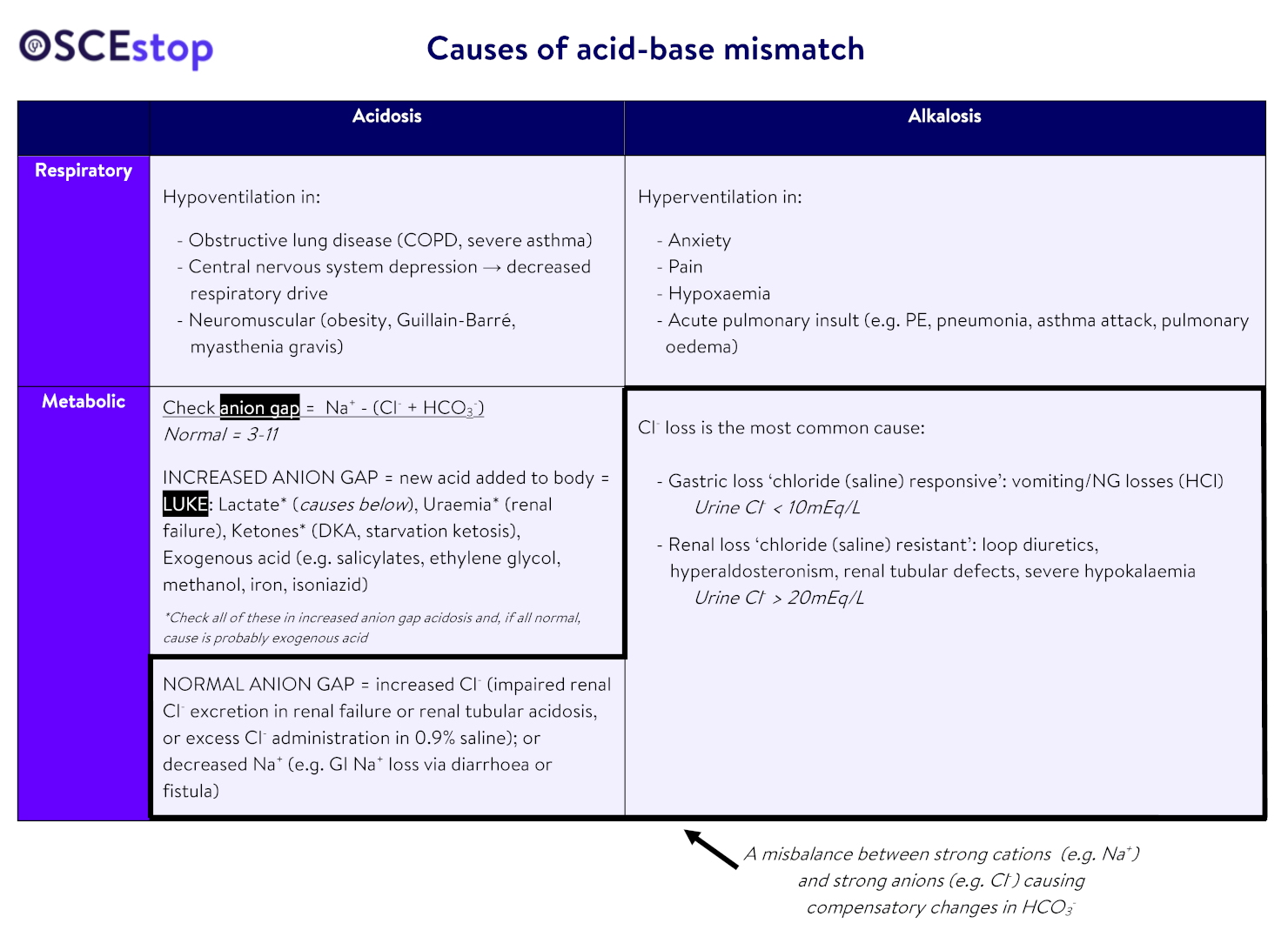
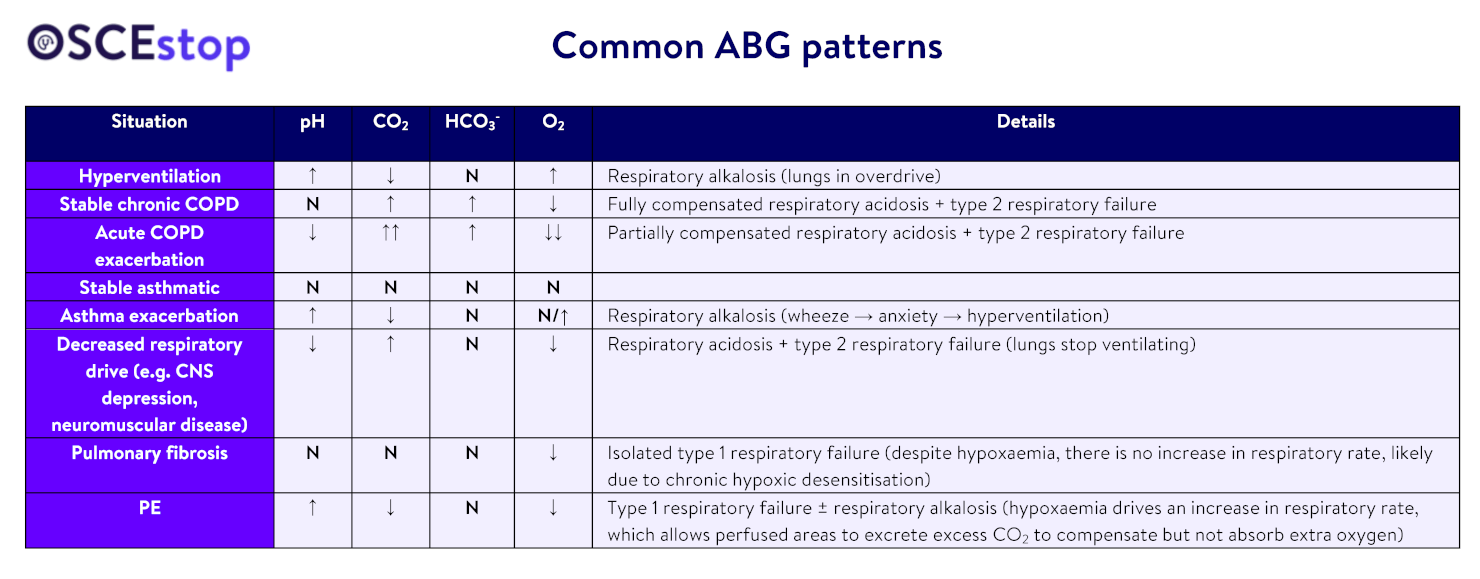


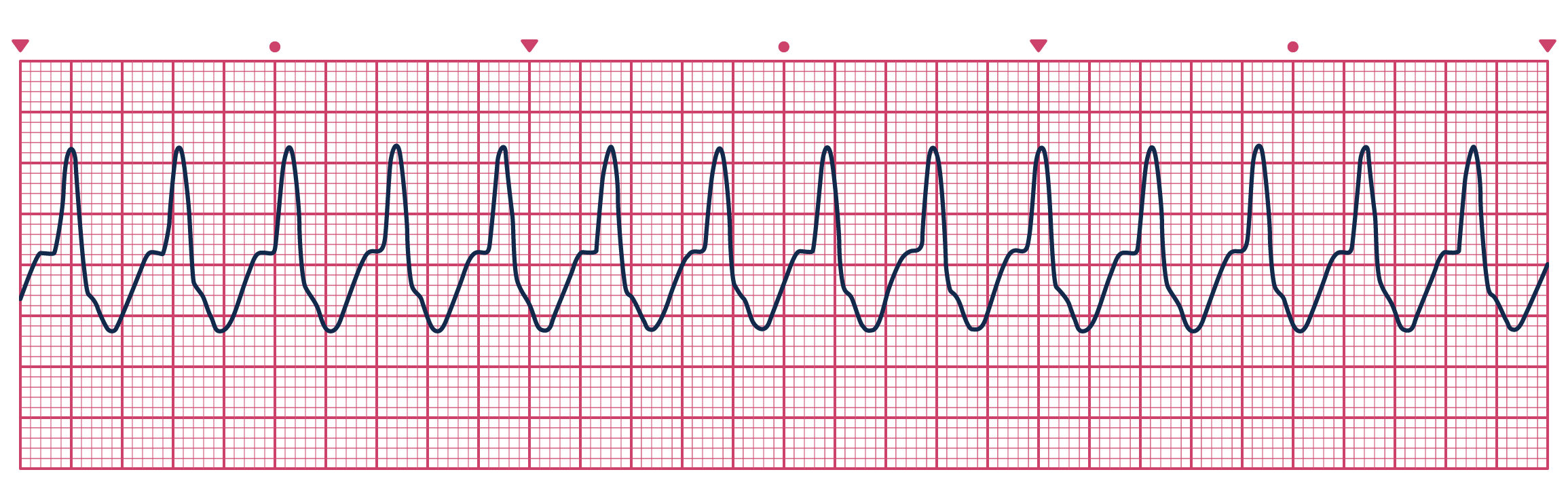
thanks
Would like to be a member
Great! You can sign up by clicking here